Chemistry
From batteries to pharma
April 9, 2026
Share
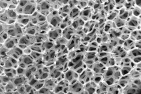
A porous piece of nickel foam, typically used in rechargeable batteries, is opening new possibilities in pharmaceutical chemistry thanks to a collaboration between Queen’s researchers (credit: Derek Esau).
For researchers, scientific discovery often means finding something new that hasn’t been observed before. But it can also involve finding a promising new use for something that’s already familiar, and this is where a new collaboration between chemistry teams at Queen’s comes in.
For more than 20 years, Gregory Jerkiewicz (Chemistry) has been working with nickel-based materials, including nickel foam. He deposits nickel on a polyurethane foam template and then burn it off. What’s left is something like a sponge, a mechanically stable, solid but very porous material.
Nickel foam is mostly used in rechargeable batteries for its light weight, chemical resistance, and porous structure – it supports active electrode materials, and together they offer large energy storage capacity and long lifetime, that is, the number of times a battery can be discharged and charged again.
A safer way to work with nickel
Dr. Jerkiewicz knew some of his colleagues in synthetic chemistry also worked with nickel, but in a different form: Raney nickel, a very fine powder, highly flammable when in contact with oxygen – and much more dangerous to handle. “I decided to give Andrew Evans (Chemistry) a call and ask if he had ever considered using nickel foam instead – he hadn’t, and we started a new collaboration in 2015”, says Dr. Jerkiewicz.
Dr. Evan’s lab focuses on exploring new metal-catalyzed reactions to prepare complex bioactive products. A postdoctoral researcher on the team, Rebecca Grange, tested various ways to use nickel foam in organic synthesis, until she discovered a promising reaction: converting nitroarenes into anilines – one of the most widely used transformations in pharmaceutical chemistry. The resulting products are common building blocks in medicines, agrochemicals, dyes, polymers, and advanced materials.
“What makes the chemistry here striking is its simplicity,” says Dr. Evans. “The reaction uses commercially available nickel foam and acidic ethanol at room temperature.” The new method can replace traditional ones, which often require pressurized hydrogen, high temperature, or specialized catalysts, all of which are more costly and generate significant waste.
Dr. Jerkiewicz calls the new process a “true breakthrough”, as it achieves high chemoselectivity – in other words, it acts in very specific parts of the molecules we want to transform, while leaving others unchanged. Even better, it does that using an Earth-abundant reagent that provides a simple, clean, room-temperature process.
Implications for pharmaceutical manufacturing
The research was published in the high-impact Journal of the American Chemical Society, and the authors are excited to share that the new method is affordable, broadly applicable and scalable, with potential impact in the pharmaceutical industry and natural product synthesis.
“This work highlights how re-examining familiar industrial materials can uncover practical solutions to longstanding synthetic challenges. In this case, a simple piece of nickel foam emerges as a surprisingly effective tool for building complex value-added molecules,” says Dr. Evans.
The project was funded by the Natural Sciences and Engineering Research Council (NSERC) Discovery Frontiers program, which granted $4 million to the Engineered Nickel Catalysts for Electrochemical Clean Energy (Ni Electro Can) research team led by Dr. Jerkiewicz.
“We expect that this research will encourage people to explore new uses of nickel and other metallic foams, sparking a new and impactful trend in synthetic organic chemistry”, says Dr. Jerkiewicz.



