University Animal Care Committee Standard Operating Procedure
Document No: 10.9.3
Subject: Intramuscular Injection in Rats
Date Issued: July 7, 2011
Revision: 4
Location: Queen’s University
Responsibility: Principal Investigators (PI), Research Staff, Veterinary Staff
Purpose: The purpose of this Standard Operating Procedure (SOP) is to describe common injection techniques used in rats.
Abbreviations:
Animal Care Services ACS, Principal Investigator PI, subcutaneous SC, intravenous IV, intraperitoneal IP, intramuscular IM, per os PO, per rectum PR
1. Introduction and Definitions:
The injection methods described within an Animal Use Protocol (AUP) must be followed at all times. The following guidelines provide recommended injection sites, needle sizes and maximum dose volumes. “Good Practices” include:
- All animals securely and safely restrained prior to injecting.
- Only three attempts per site should be practiced. If unsuccessful, allow another (trained and competent) person to collect the sample.
- Use the appropriate gauge needle and volume for the injection site based on the size of the rat.
- Before injecting any substance, aspirate first to ensure appropriate placement of the needle (excluding intravenous injections).
- Always inject with the bevel up on the needle.
- Always ensure the substances you are injecting are sterile, and use sterile technique.
- Every animal requires a new sterile syringe and a new sterile needle. With small volumes, it is preferable to dilute the injectable agent to a 50% or less solution to ensure accurate dosing.
- Disinfecting the skin with alcohol is mandatory for intravenous, intraperitoneal, intramuscular, intradermal, and all biohazardous injections.
- Choose the appropriate administration route for the substance to be injected.
| Recommended Needle Sizes and Volumes Length of needle: 1/2 to 3/4 inch |
|
|---|---|
| Intramuscular IM | |
| Recommended Gauge (maximum) |
26 (25) |
| Good Practice Volume (Max per site) |
0.1 ml |
2. Materials:
- Sterile syringes
- Sterile needles (21-27 gauge)
- 70% alcohol
- Injectable solution
- Anesthetics as required (if using anesthetics: eye lubricant, heating source, Isotonic fluids – see SOP 10.6 Anesthesia in Rats)
3. Procedures:
Anatomical Terms of Location
- Every animal requires a new sterile syringe and a new sterile needle.
- Load the syringe and needle with appropriate volume to be injected.
- Safely restrain the animal using either physical or chemical restraint.
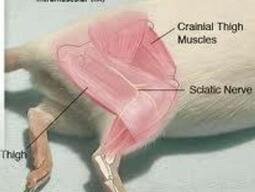
- Place your hand on the inside of the animal’s hind leg and gently extend the leg, stabilizing the muscle.
- Palpate the hamstrings on the caudal aspect of the femur.
- Disinfect the injection site with 70% alcohol.
- Care must be taken to avoid injecting material near the sciatic nerve which runs superficially along the caudal aspect of the femur in the thigh. The needle must be directed caudally with the bevel up.
- Aspirate to ensure the placement of the needle is correct. Proper placement should yield negative pressure and no aspirate in the hub of the needle. If any fluids are seen; stop, reload with new syringe and needle, check injection site for trauma, reposition needle and attempt again.
- After ensuring proper placement, inject.

| Date | New Version |
|---|---|
| 07/07/2011 | Created |
| 01/24/2012 | Annual Review |
| 09/22/2015 | Triennial Review |
| 02/28/2019 | Triennial Review |
| 02/28/2022 | Triennial Review |
| 12/12/2022 | Triennial Review, split apart separate injection methods, updated format and reviewed/updated maximum injection volumes |
 Research at Queen's
Research at Queen's
