University Animal Care Committee Standard Operating Procedure
Document No: 7.25
Subject: Estrus Cycle Monitoring (Mice)
Date Issued: March 24, 2017
Revision: 2
Location: Queen’s University
Responsibility: Principal Investigators (PI), Research Staff, Veterinary Staff
Purpose: The purpose of this Standard Operating Procedure (SOP) is to describe common techniques used for estrus monitoring in mice.
Abbreviations:
Animal Care Services ACS, Principal Investigator PI, subcutaneous SC, intravenous IV, intraperitoneal IP, intramuscular IM, per os PO, per rectum PR
1. Introduction and Definitions:
Microscopic evaluation of the types of cells present in vaginal smears has long been used to document the stages of the estrous cycle in laboratory mice. Their estrous cycle averages 4–5 days and is a repetitive but dynamic process whereby different cell types appear and recede in waves throughout the cycle, reflecting changes in the levels of estradiol and progesterone secreted by the ovarian follicles. The estrous cycle is generally divided into the four stages of proestrus, estrus, metestrus, and diestrus. On cytological evaluation, these stages are defined by the absence, presence, or proportion of four basic cell types as well as by the cell density and arrangement of the cells on the slide. Vaginal cytology can be evaluated immediately after collection as an unstained, wet mount preparation (direct cytology) or as a fixed and stained slide preparation.
2. Materials:
- Pipette and sterile tips (recommended 1.5 mm internal tip bore) OR
- Disposal sterile transfer pipettes
- Sterile physiological saline (NaCl)
- Sterile swabs
- Glass slides (plain, or commercially available pre-drawn numbered grid)
- Coverslips
- Metachromatic stains (Romanowsky-type stains (e.g., Modified Wright’s, Wright’s Giemsa) and Toluidine Blue O are the commonly used cytological stains)
** A new tip, disposal pipette or swab must be used for each and every animal to prevent between-animal sample contamination that may lead to inaccuracies in staging or infection of the reproductive system. **
3. Procedures
- To elucidate the length and, subsequently, the stages of the estrous cycle, vaginal cytology samples are collected over at least 14 consecutive days. Vaginal cytology samples can be collected at any time of the day, but the common practice is to collect them early in the morning after the lights are turned on. Regardless of when samples are collected, it should be done at approximately the same time of the day over the course of the collection period to reduce variability. Collection of the samples can be done either by vaginal lavage or swabbing. Lavage typically yields a higher cellularity sample, and minimizes cellular distortion. Regardless of the methodology employed, accurate interpretation of vaginal cytology samples is dependent on the quality of the sample preparation.
- Load the pipette with 100uL of ambient temperature, sterile NaCl.
- Gently remove the female from the home cage and place on a clean, flat surface. Or on the wire lid.
- Using the base of the tail, slightly elevate the hind end. Alternatively, hold the animal securely in the scruff hold (see SOP 7.20 “Manual Restraint of Mice”).
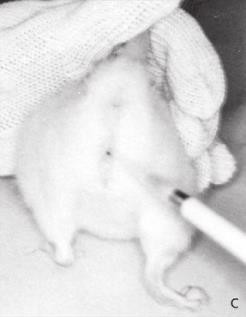
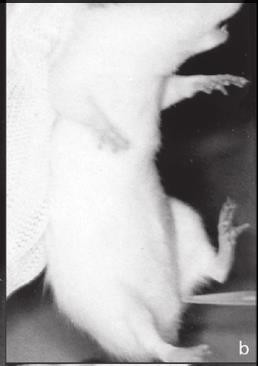
- Gently insert the tip of the pipette into the vaginal orifice at a depth of approximately 1–2 mm.
- Flush the NaCl into the vagina and back out 2 or 3 times. If after the first flush the fluid is cloudy, subsequent flushings may not be necessary.
- When inserting and flushing into the vaginal orifice, care must be taken not to insert the tip too deep so as to avoid cervical stimulation, which can induce pseudo-pregnancy and appears as a persistent diestrus for up to 14 days.
- Mark the animal (if group housed), record the procedure on the cage card, and return her to the home cage.
- After the lavage, a small drop of the sample is then placed evenly on the slide in a thin layer (smear) and allowed to air dry.
- Crystals may form on the dry unstained slides; these will be removed as part of the staining process. It is important to avoid heavy staining and to rinse the slides thoroughly.
- Dry fixed slides do not require a spray fixative prior to staining. A spray fixative is used only when a stain requires wet fixation.
- Alternatively, a vaginal swab using a sterile swab and ambient temperature NaCl may be used. While gently restraining the animal, rotate the swab gently and roll against the vaginal wall.
- Remove the swab, mark the animal (if group housed), record the procedure on the cage card, and return her to the home cage.
- Transfer the cells to a dry glass slide by rolling the swab across it. Air dry and stain.
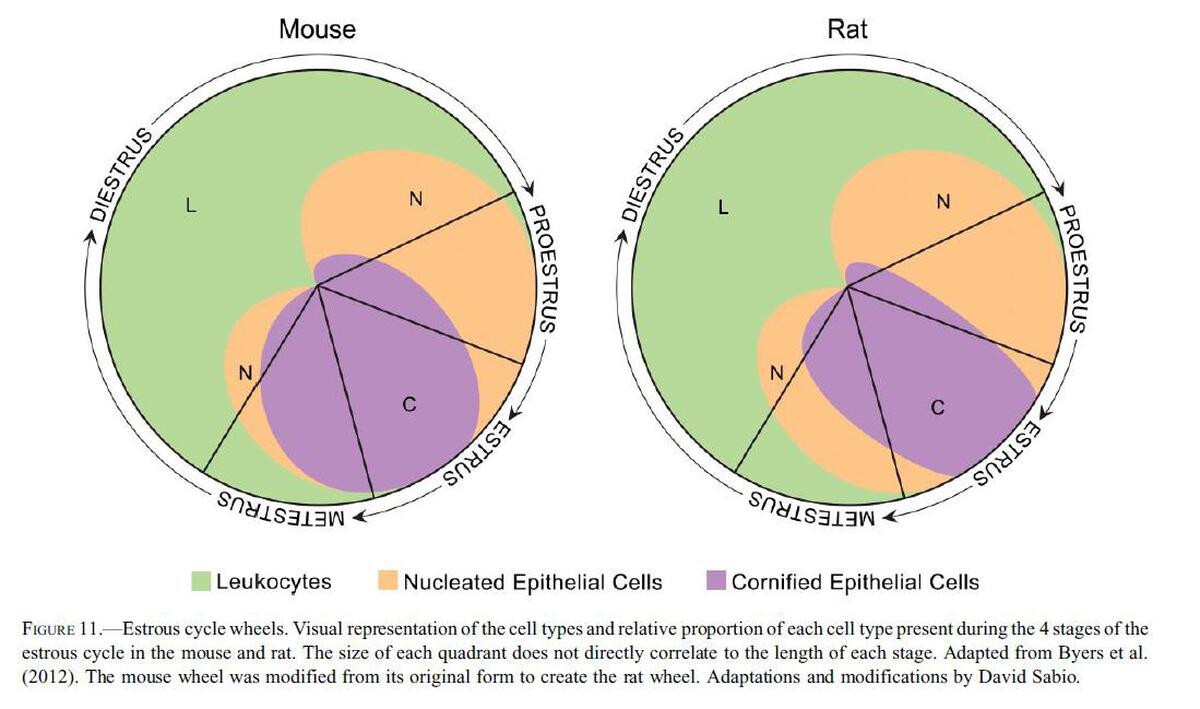
The Cells and Stages of the Estrous Cycle
Estrous cycle length averages 4–5 days in mice. Many factors influence cycle length including light, age, temperature, noise, nutrition, stress, and social relationships. Female rodents cycle more regularly when males are present in the room (Whitten effect). The length of the four stages varies between 6 and 72 hr depending on the stage and individual rodent, therefore, some short stages may be ‘‘missed’’ especially if samples are collected very early or late in the day. For example, proestrus, with an average length of 14 hr, could be missed with an early morning sample collection because in many females proestrus does not start until midmorning and would end before the next collection. Sample collection can be shifted to late morning or early afternoon in an effort to minimize the incidences of missed stages.
The Cells of the Estrous Cycle
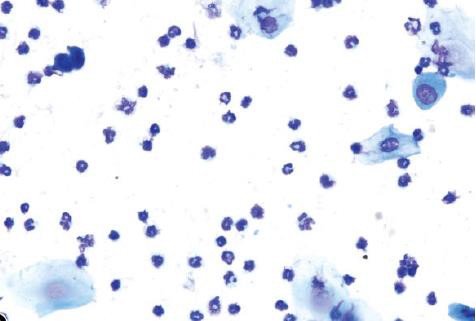
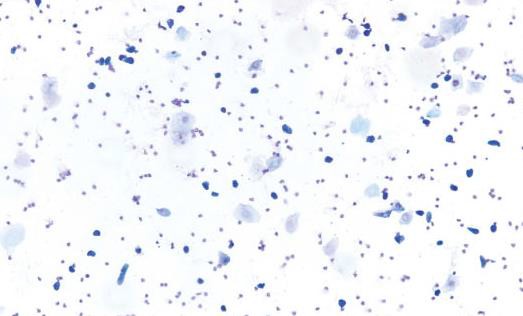
Neutrophils
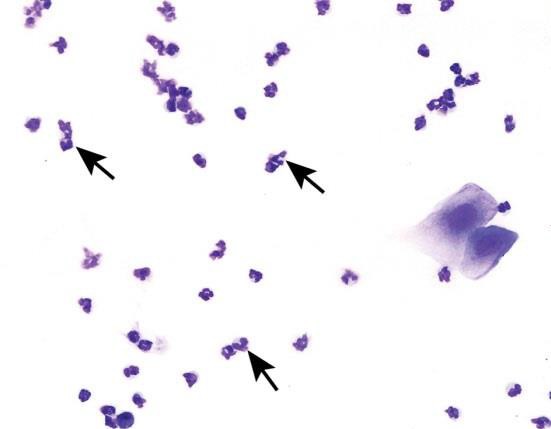
Neutrophils are also known as leukocytes or polymorphonuclear cells. These cells are round, very small, and possess multilobulated nuclei. Neutrophils are relatively delicate cells and can sometimes rupture during collection or processing. It is important to recognize the appearance of ruptured neutrophils to avoid misinterpretation of a particular stage.
Small Nucleated Epithelial Cells

These cells are small, round to oval, and nonkeratinized. They have a high nuclear to cytoplasmic ratio relative to large epithelial cells, a round nucleus, and blue cytoplasm. They may stain very dark or basophilic, precluding visualization of the nucleus. Small cytoplasmic vacuoles may be seen in occasional small epithelial cells of proestrus.
Large Nucleated Epithelial Cells

These cells are larger with moderate to abundant amounts of blue to sky blue cytoplasm and a lower nuclear to cytoplasmic ratio in comparison to small epithelial cells. They are round to polygonal and may have irregular, jagged, or angular borders. Large epithelial cells can have some degree of keratinization and possess nuclei that may be intact, degenerate, or pyknotic.
Occasionally, large nucleated epithelial cells will contain finely stippled cytoplasmic granules.
Anucleated Keratinized Epithelial Cells
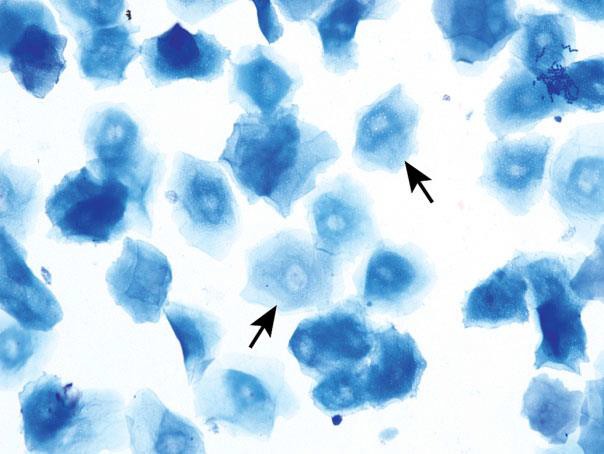
Anucleated epithelial cells are also known as squames or ‘‘cornflakes.’’ They are aged cells and are characterized by an abundant blue to sky blue cytoplasm with jagged or angular edges. They lack nuclei, although pale round areas representing where a nucleus had existed (nuclear ‘‘ghost’’) may be visualized. The distal one-fourth to one-third of the vaginal lumen is covered by squamous epithelial cells and is permanently keratinized.
Stages of the Estrous Cycle
The stages of the estrous cycle are identified by the absence, presence, or proportion of the described four basic cell types as well as by the cell density and arrangement of the cells on the slide. Conventionally, the cycle is divided into the four stages of proestrus, estrus, metestrus, and diestrus, however some researchers condense or subdivide the stages.
One general approach to staging vaginal smears is to first assess for the presence of neutrophils. If neutrophils are a dominant feature, or consistently observed, the stage is either metestrus or diestrus; if neutrophils are rare to absent, the stage is either proestrus or estrus.
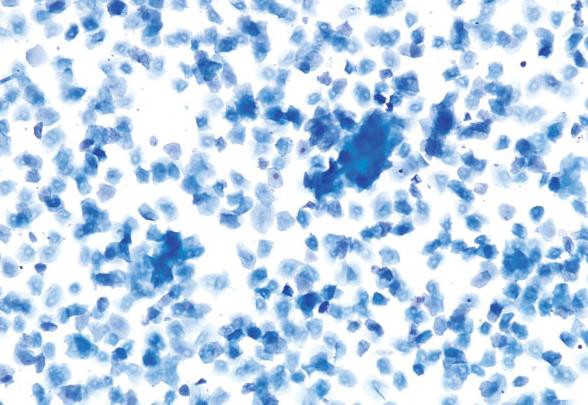
Proestrus
Proestrus is a short stage, lasting an average of less than 24hr in mice. The predominant feature of this stage is the presence of small, round, nucleated epithelial cells of relatively uniform appearance and size. Typically, no neutrophils will be seen. That said, rare to occasional neutrophils can be found in early proestrus as the rodent would have recently transitioned from diestrus into proestrus.
Relatively low numbers of large epithelial cells and keratinized anucleated cells may also be observed. As the cycle approaches estrus, keratinized cells will become more abundant. The presence of low numbers of neutrophils, or large and anucleated epithelial cells, does not preclude the diagnosis of proestrus when the predominating feature of the smear are the small, round epithelial cell population.

Estrus
Estrus duration ranges between 12 and 48hr in mice. It is characterized by the presence of predominately anucleated keratinized epithelial cells. Numerous bacteria may be observed adhered to the cells or free in the background. Occasional nucleated epithelial cells can be observed throughout the stage of estrus, and neutrophils are absent or occasionally observed in late estrus. The first and second half of estrus, while consisting mostly of anucleated keratinized cells, have distinct differences in their appearances and also differ between rats and mice. In mice, at the start of estrus, the anucleated cells are smaller and are typically arranged in loose clusters or sheets reminiscent of proestrus.

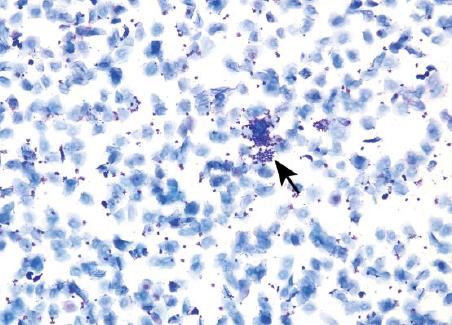
Metestrus
Based on the limited reports in literature the duration of metestrus in mice seems to last up to 24 hr and, although infrequent, metestrus has been identified for 2 consecutive days in control mice on reproductive studies. Metestrus is characterized by a combination of anucleated keratinized epithelial cells and neutrophils. In mice, nucleated cells may appear occasionally throughout metestrus.
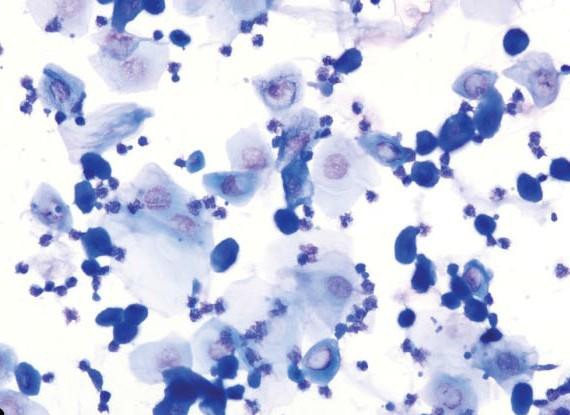
The separation between where metestrus ends and diestrus begins is not always obvious, as they (i.e., the end of metestrus and start of diestrus) are very similar in appearance and are defined by the same cell types.
Early and mid metestrus, however, are easily identified if and when sample collection happens to occur at these times.
Diestrus
Diestrus is the longest stage of the estrus cycle with an average duration of 48–72 hr in mice. This stage is characterized by a substantial decrease in the number of anucleated keratinized epithelial cells (but not necessarily an absence of) as the animal transitions out of metestrus. Overall cellularity is moderate to low with a combination of neutrophils, small and large nucleated epithelial cells, and low numbers of anucleated keratinized cells. Neutrophil numbers can vary but are usually higher in number relative to the epithelial cells with smears sometimes being exclusively neutrophilic. Occasionally, in early diestrus, neutrophils may still appear in clumps. It is not unusual for diestrus smears to have a very low cellularity, especially on day 2 or 3 of diestrus, with only a sparse scattering of neutrophils and epithelial cells. In late diestrus, the epithelial cells may become more round or be organized in small clumps, indicating proestrus the next day; however, neutrophils will still be consistently observed. During diestrus, macroscopic vaginal excretions are low.
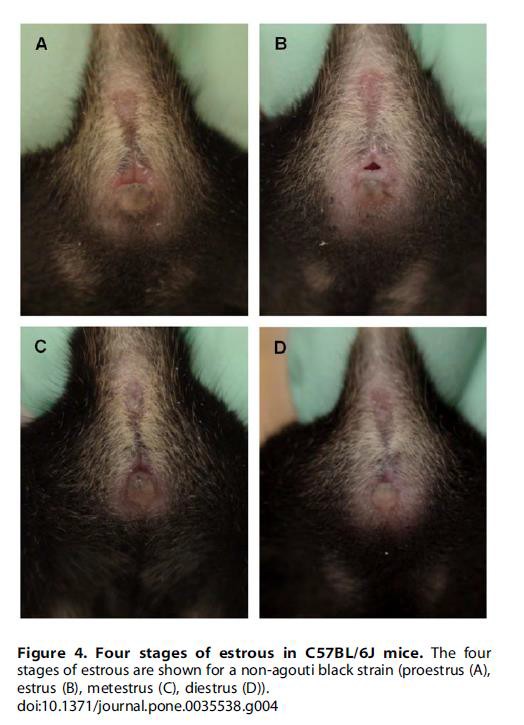
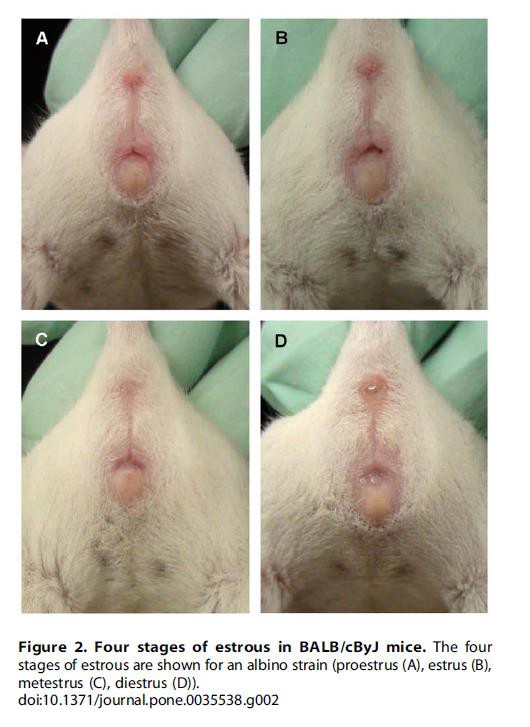
It is worth noting that changes occurring in the mouse estrous cycle are evident in the animal’s physiology and anatomy. With practice, using a method for determining the stage of estrous by visual observation may be quicker than the cytological methods although not as precise. Until skilled in this, it is recommended that lavage or swabbing continue as standard practice.
Vaginal Cytology of the Laboratory Rat and Mouse: Review and Criteria for the Staging of the Estrous Cycle Using Stained Vaginal Smears Michelle c. Cora 1, l=Linda Kooistra 2, and Greg Travlos. Cellular and Molecular Pathology Branch, National Toxicology Program, National Institute of Environmental Health Sciences, National Institutes of Health, Research Triangle Park, North Carolina, USA 2Charles River Laboratories, Inc., Pathology Associates, Durham, North Carolina, USA
Determination of the estrous cycle phases of rats: some helpful considerations. Marcondes, F. K., bianchi, f. j. and Tanno, A. P. Depto. Ciências Fisiológicas, Faculdade de Odontologia de Piracicaba, Universidade Estadual de Campinas, Av. Limeira, 901, CEP 13414-903, Piracicaba, SP, Brazil
A quantitative method for assessing stages of the rat estrous cycle.CH Hubscher, DL Brooks, JR Johnson Department of Anatomical Sciences & Neurobiology, University of Louisville, Louisville, Kentucky 40292 Submitted June 17, 2004; revised February 2, 2005; accepted March 8, 2005
Mouse Estrous Cycle Identification Tool and Images. Shannon L. Byers1, Michael V. Wiles2, Sadie L. Dunn1, Robert A. Taft11 Reproductive Sciences R&D, The Jackson Laboratory, Bar Harbor, Maine, United States of America, 2 Technology Evaluation and Development, The Jackson Laboratory,Bar Harbor, Maine, United States of America
| Date | New Version |
|---|---|
| 03/24/2017 | Created |
| 01/28/2020 | Tri Annual Review |
| 02/06/2023 | Tri Annual Review and update format |
 Research at Queen's
Research at Queen's